Worksheets for Fractional Distillation Process Of Crude Oil
Fractional distillation is used in oil refineries (Figure 5.41) to separate the complex mixture into fractions that contain similar boiling points and therefore similar molecular weights and properties. Gasoline, diesel fuel, kerosene, and jet fuel are some of the different fractions produced by an oil refinery.

Fractional Distillation Of Crude Oil Diagram HighRes Vector Graphic Getty Images
Main Processing Units of Refineries - Treatment Crude Oil Distillation unit: Distills the incoming crude oil into various fractions for further processing in other units. Vacuum distillation unit: Further distills the residue oil from the bottom of the crude oil distillation unit.
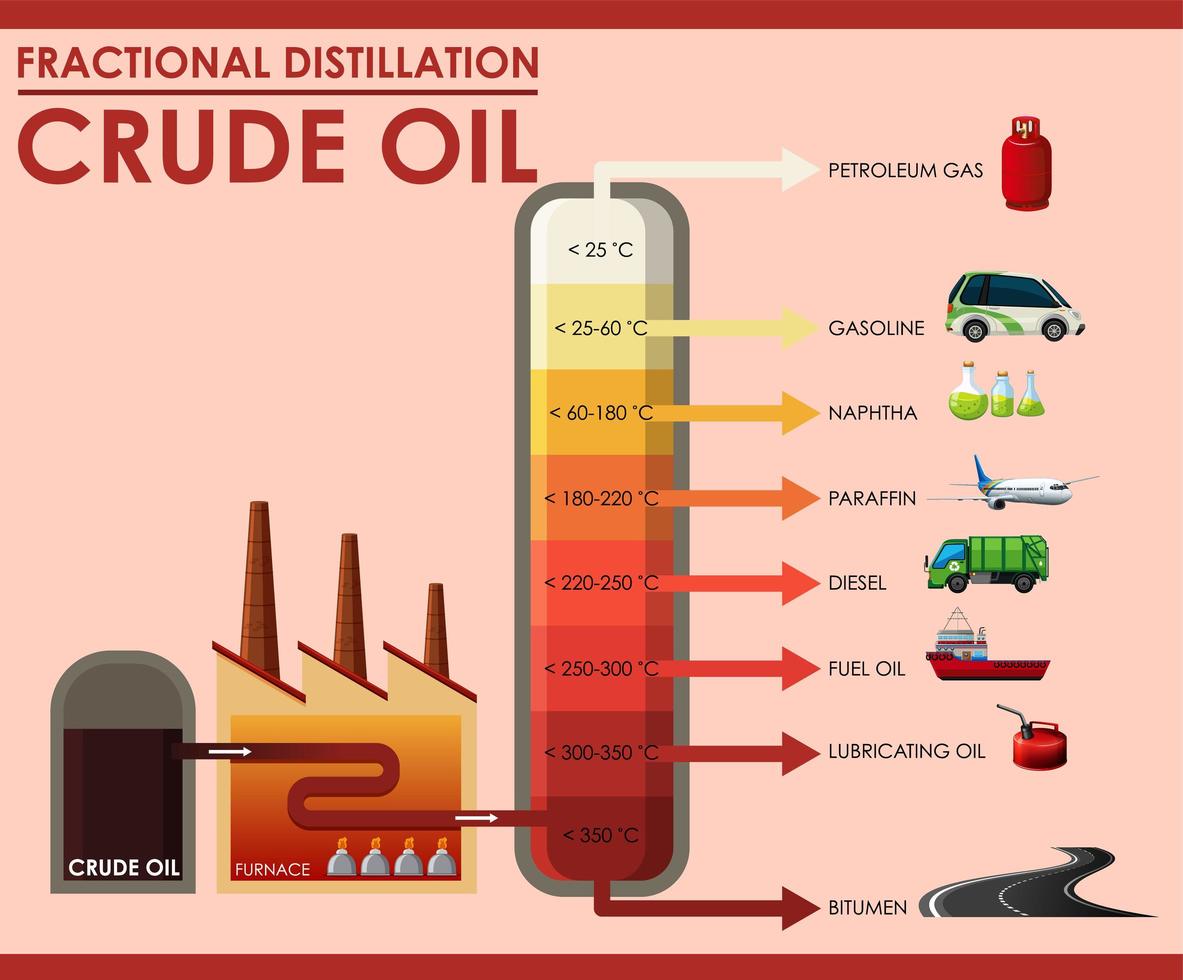
Diagram showing fractional distillation crude oil 1235260 Vector Art at Vecteezy
Fractional distillation is the primary means of separation of crude oils at refineries into fractions of different boiling ranges. The fractions are subsequently upgraded or converted into useful products in other units. For very heavy fractions, extraction becomes the method of choice for further separation.

What is Fractional Distillation? The Chemistry Blog
Fractional distillation separates a mixture into a number of different parts, called fractions. A tall fractionating column is fitted above the mixture, with several condensers coming off at.

How fractional distillation of crude oil works. Fractional distillation, Crude oil, Petroleum
In this Online Class, your favourite master teacher Nabamita will teach Fractional Distillation of Petroleum of Class 8 through this live session. Learn more.
Crude Oil The Fractional Distillation Of Crude Oil
Fractional distillation is a type of distillation which involves the separation of miscible liquids. The process involves repeated distillations and condensations and the mixture is usually separated into component parts. The separation happens when the mixture is heated at a certain temperature where fractions of the mixture start to vaporize.
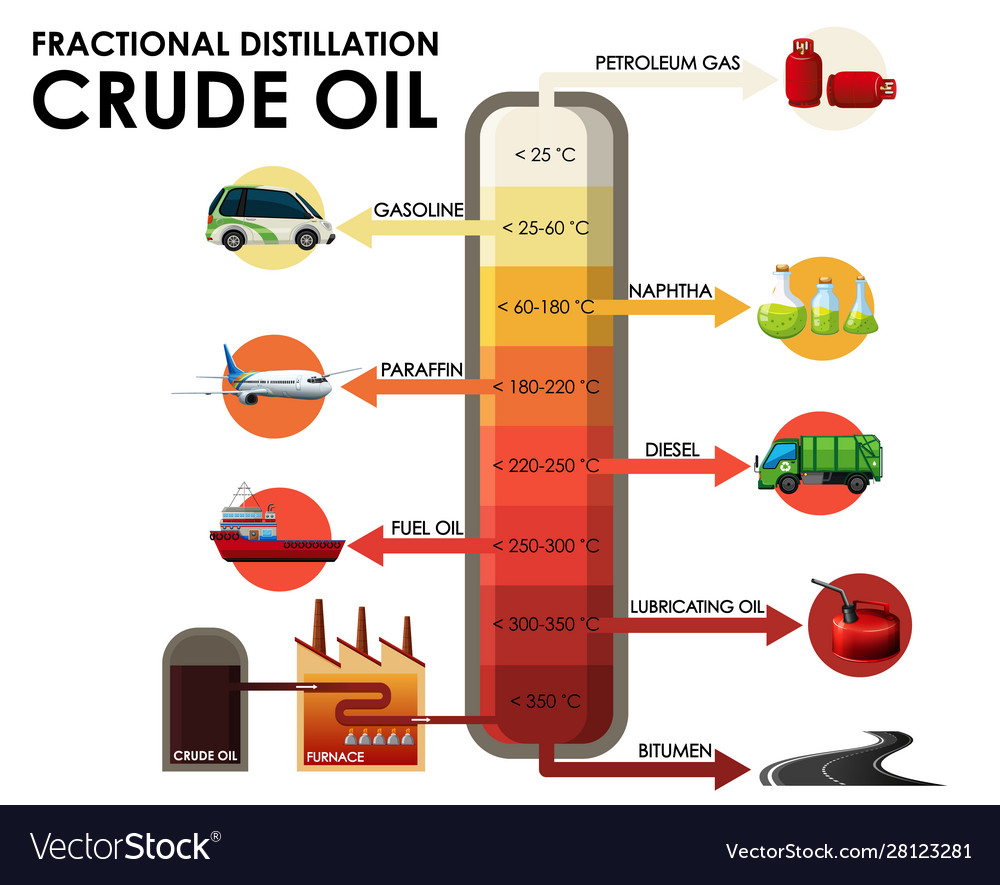
Diagram showing fractional distillation crude oil Vector Image
This is done by a process called fractional distillation which is based on the fact that the different components of petroleum have distinctly different boiling points. In fractional distillation, crude petroleum is heated to a temperature of, 400°C or slightly above in a furnace.
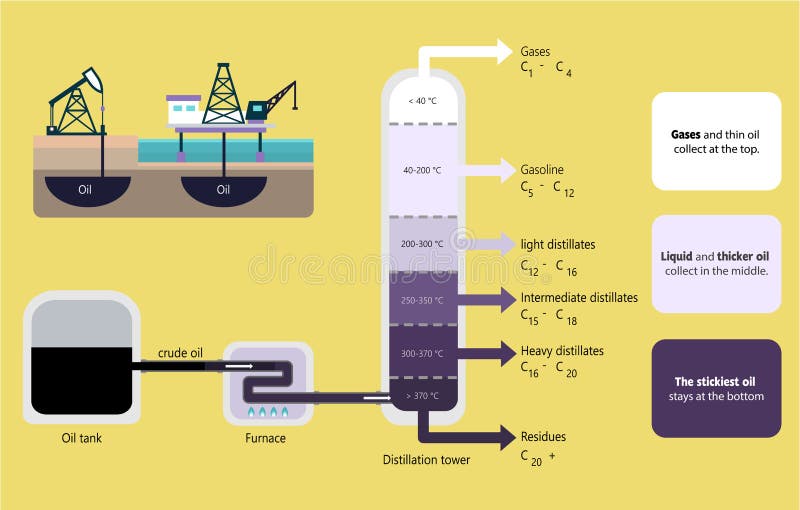
Fractional Distillation of Crude Oil Diagram Stock Vector Illustration of petroleum, crude
The fractions in petroleum are separated from each other in a process called fractional distillation The molecules in each fraction have similar properties and boiling points, which depend on the number of carbon atoms in the chain The size and length of each hydrocarbon molecule determines in which fraction it will be separated into

Diagram showing fractional distillation crude oil Vector Image
It is assumed that readers have previously performed a simple distillation, so in this section are described differences between simple and fractional distillation. Figure 5.43: Fractional distillation apparatus. Figure 5.44: a) Removal of glass wool plug on a beaded fractionating column, b) Insulating the column with foil, c+d) Condensation on.

Generic Fractional Distillation in the Crude Oil Refining Process. Download Scientific Diagram
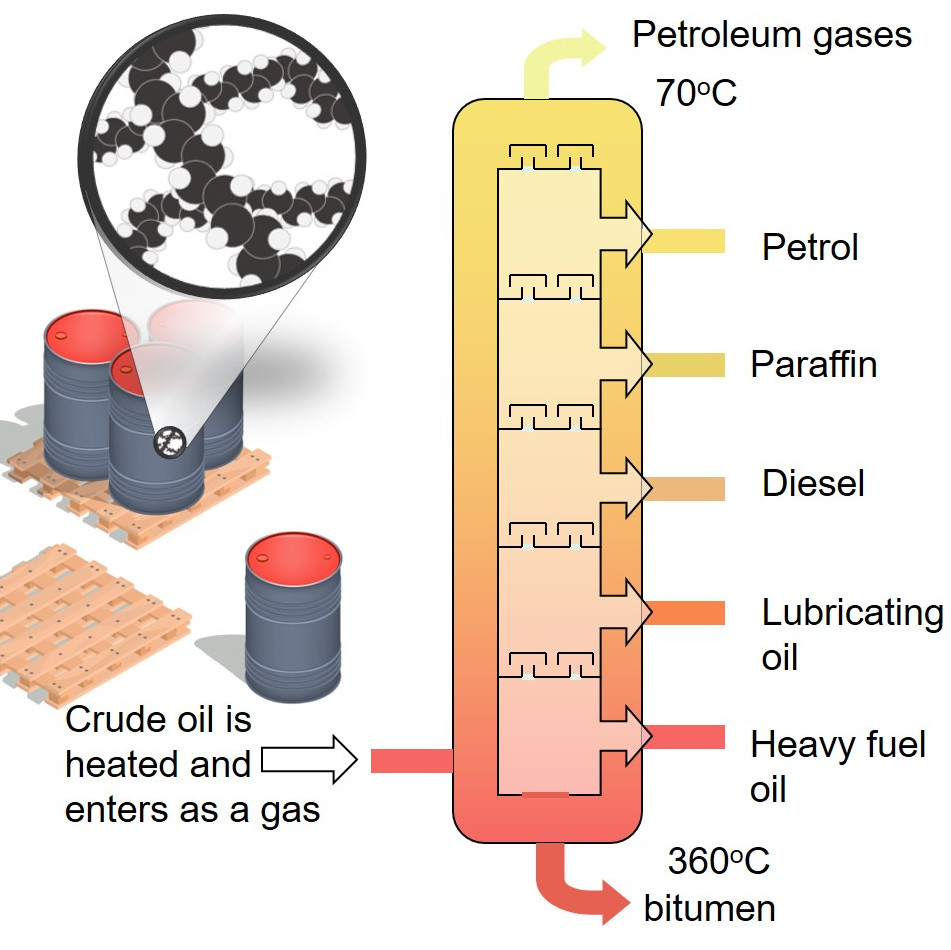
Fractional distillation separates crude oil into fractions - groups of hydrocarbon molecules with similar carbon chain lengths, properties and boiling points. Each fraction has a different use. Did you know.? The oil tankers that carry crude oil to refineries are some of the largest ships and can be over 350 metres long.
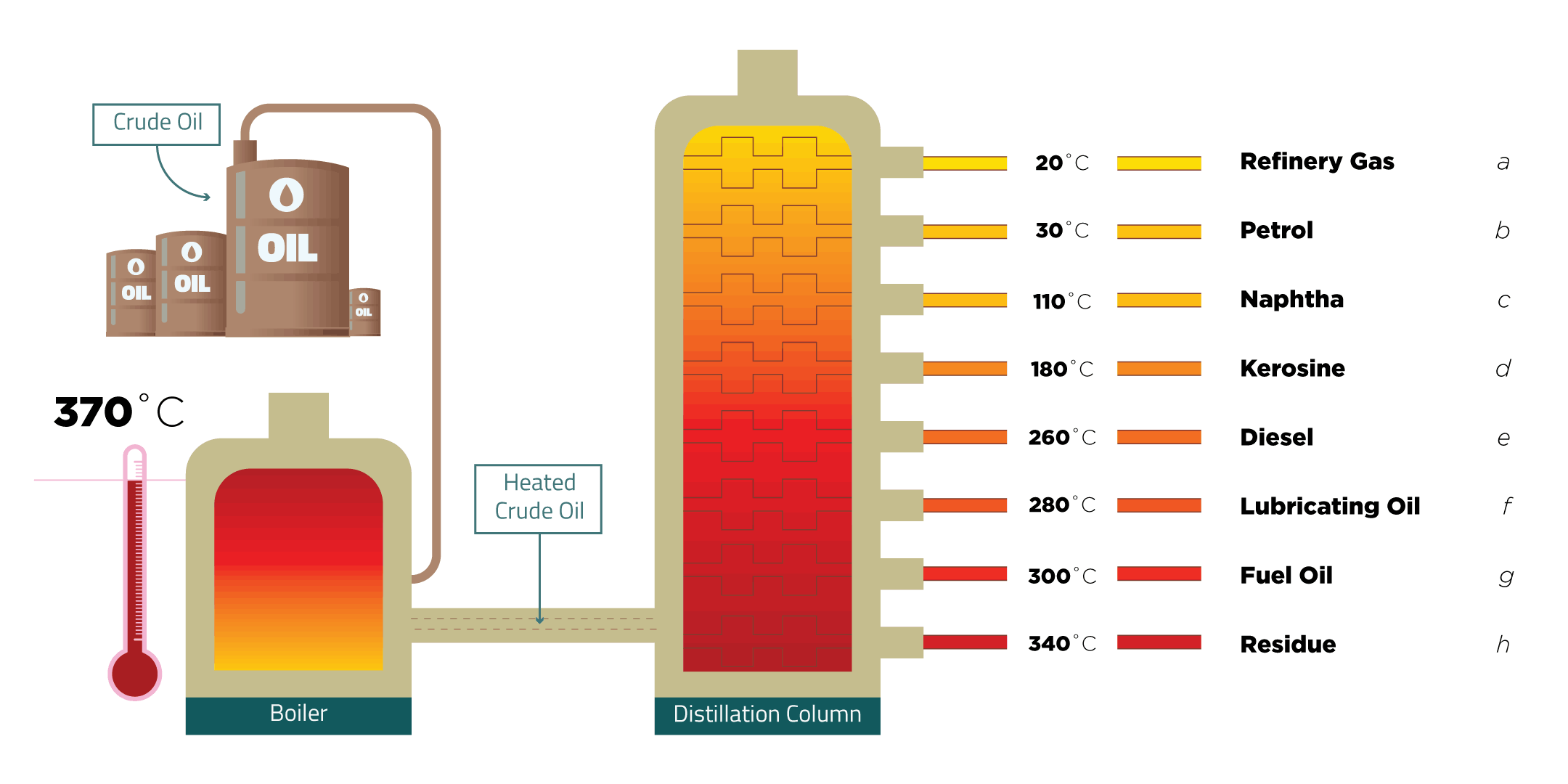
Crude Oil The Fractional Distillation Of Crude Oil
Crude oil as a mixture is not a very useful substance but the different hydrocarbons that make up the mixture, called fractions, are enormously valuable, with each fraction having many different applications. Diagram showing the process of fractional distillation to separate crude oil in a fractionating column. Fractional distillation is.
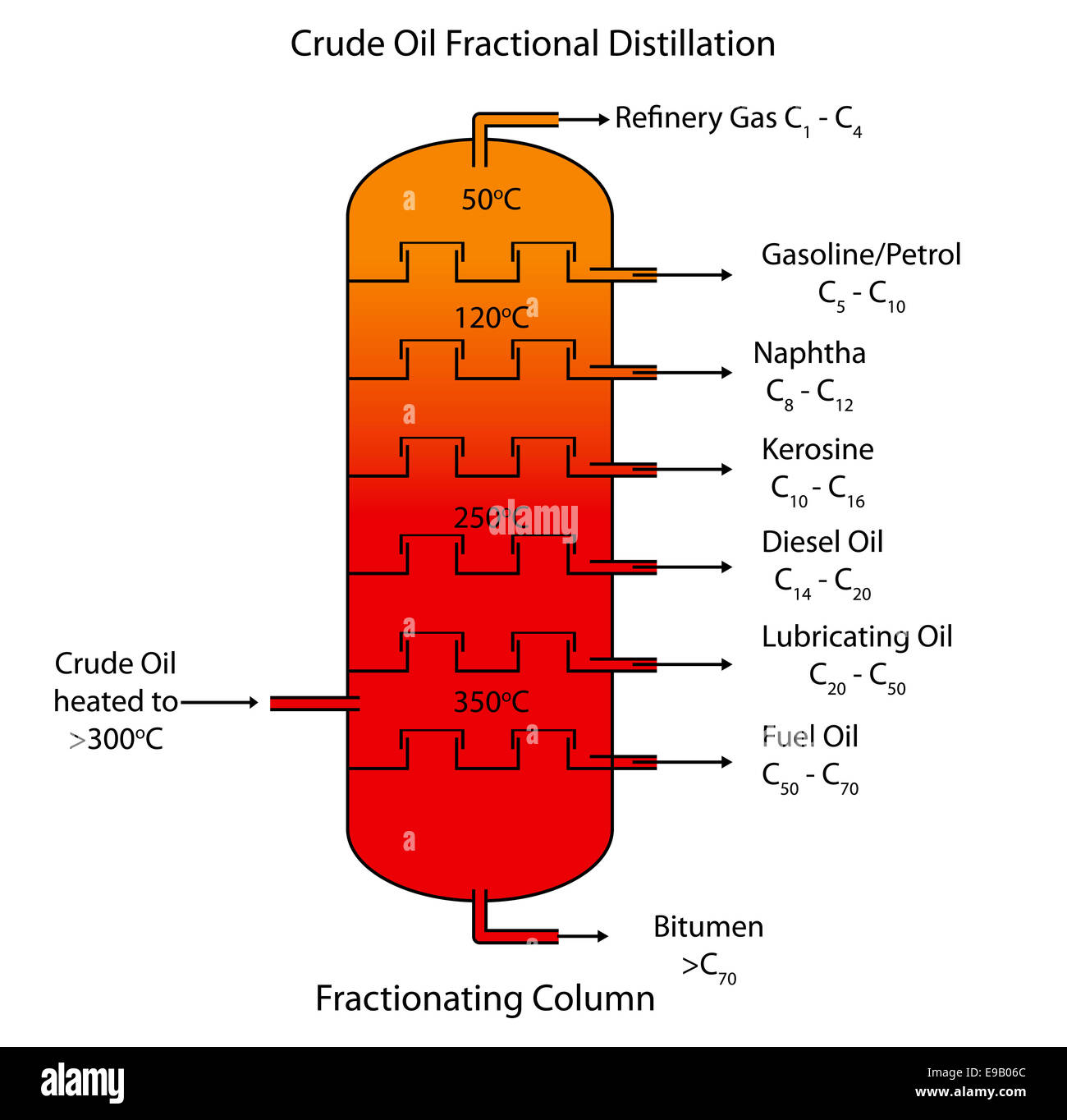
Labeled diagram of crude oil fractional distillation Stock Photo, Royalty Free Image 74615028
Relating what happens in the fractionating column to the phase diagram. Suppose you boil a mixture with composition C 1. The vapor over the top of the boiling liquid will be richer in the more volatile component, and will have the composition C 2. That vapor now starts to travel up the fractionating column.

Diagram showing fractional distillation crude oil Vector Image
Download PDF Did this video help you? Fractional Distillation of Crude Oil Crude oil as a mixture is not a very useful substance but the different hydrocarbons that make up the mixture, called fractions, are enormously valuable, with each fraction having many different applications

Crude oil fractional distillation labeled diagram Vector Image
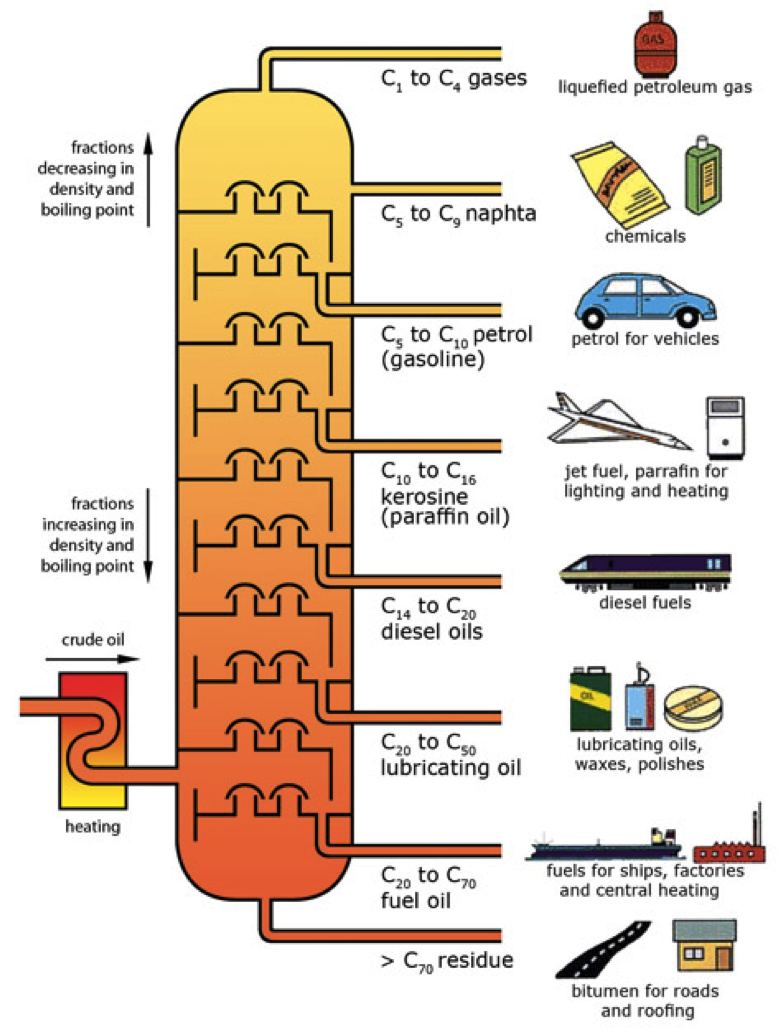
Figure 1. Diagram of a fractional distillation tower, showing where the different fractions will condense. [1] Note that the temperature is higher at the bottom, so the longer carbon chains will fall out at the bottom, the shorter carbon chains will go up the column until they hit a temperature at which they become liquid.

Crude Oil The Fractional Distillation Of Crude Oil
In the petroleum industry, fractional distillation is used for refining crude oil where the components have similar boiling points. Crude oil can be separated by fractional distillation into useful products: liquefied petroleum gas, gasoline, kerosene, jet fuel, diesel oil and fuel oils. The products of crude oil include many
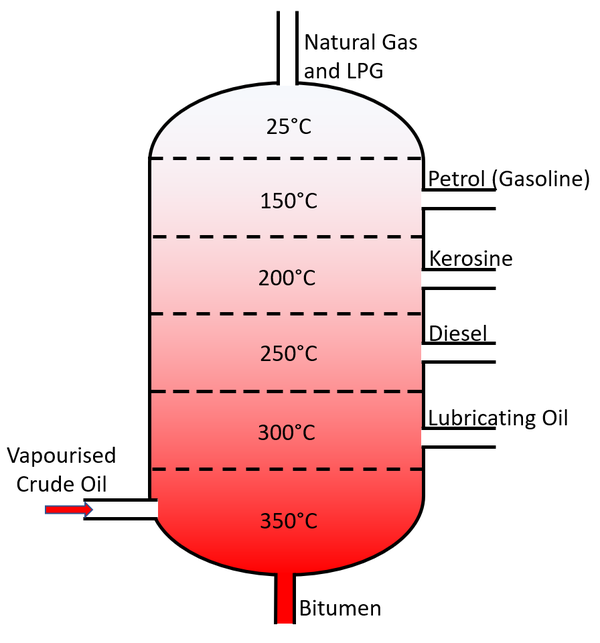
Fractional Distillation Key Stage Wiki
Gasoline. Petroleum is converted to useful products such as gasoline in three steps: distillation, cracking, and reforming. Recall from Chapter 1 that distillation separates compounds on the basis of their relative volatility, which is usually inversely proportional to their boiling points. Part (a) in Figure 3.8.1 shows a cutaway drawing of a column used in the petroleum industry for.